
I’ve read somewhere that COVID is eventually going to infect us all, but I had begun to doubt that it would catch me.
Until it did.
It’s been more than 2-1/2 years since our world was shook when the pandemic washed over us, beginning locally when the Utah Jazz-Oklahoma City Thunder game was cancelled on March 11, 2020.
That was a jarring, scary night, with both my wife and my mother in-law at that game.
Anyway, we masked up and stayed home much of 2020. Then just after Christmas of that year my wife had a mild COVID infection. She tested positive two weeks in a row.
I tested at the same time and was negative each time.
Also, I received the Pfizer vaccine in early January 2021 and declared myself bullet proof, even at my advanced age of 69. I’ve added two booster shots since then.
Then a little over a week ago I woke in the middle of the night with an unexpected, out-of-the-blue sore throat. No big deal.
But I lost energy and appetite throughout the next day, while adding a cough and tons of drainage.
By the third day, it moved into my chest and I had no taste or smell.
So, I went to a walk-in clinic. I was tested for STREP, Flu and COVID. The COVID test came back positive, which I was NOT expecting.
The doctor gave me some pills to take for the next five days, as well as a nasal spray. He said I should no longer be contagious after five days.
Things didn’t improve over the next four days, so by Sunday I walked into the Mercy Health ER along I-35 to see if I could find some relief. The health care professionals there were awesome and empathetic while giving me a steroid shot and hydrating IV.
By Monday, there was incredible improvement in my condition. I’m still improving and hope to be completely symptom free by the end of this week.
Meanwhile, what of my wife, who sleeps and eats with me? She had laryngitis while all of this was going on, and has been tested twice for COVID over the past week.
Negative, both times. So, it’s role reversal this time.
I’ve learned my lesson. I’m not bullet proof. I am thankful for the vaccines and boosters. Because of my age and medical history, there’s no telling how far down COVID could have taken me.
Now I’m up for the Bivalent booster in a couple weeks. I’m all in.



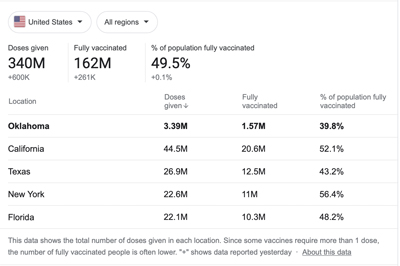 I just peeked at the numbers, and while 46 percent of my fellow Oklahomans have received at least one dose of the vaccine, less than 40 percent of us have been fully vaccinated against COVID.
I just peeked at the numbers, and while 46 percent of my fellow Oklahomans have received at least one dose of the vaccine, less than 40 percent of us have been fully vaccinated against COVID.
 FUN AT THE OL’ BALLYARD: The Dodgers have a fun bit between innings late in the game where fans dance (mostly) badly and cameras broadcast their (lack of) talent on the giant hi-def scoreboard screen. There were the usual kids dancing wildly, girls and then a couple of 20-something guys who suddenly ripped off their T-shirts like they were World Wide Wrestling contestants. Laughter erupted throughout the stands. Later, I spotted the shirtless guys sitting behind the first base dugout and giving the umpires the business. Must have been dollar beer night.
FUN AT THE OL’ BALLYARD: The Dodgers have a fun bit between innings late in the game where fans dance (mostly) badly and cameras broadcast their (lack of) talent on the giant hi-def scoreboard screen. There were the usual kids dancing wildly, girls and then a couple of 20-something guys who suddenly ripped off their T-shirts like they were World Wide Wrestling contestants. Laughter erupted throughout the stands. Later, I spotted the shirtless guys sitting behind the first base dugout and giving the umpires the business. Must have been dollar beer night. FIREWORKS: The opening night game was followed by a spectacular opening night fireworks show that was incredibly loud. There are two hotels located just over the left field fence. The game ended about 10:30 p.m. Can you imagine the unexpected jolt that sleeping patrons received? Followed by angry calls to the front desk, I’m sure.
FIREWORKS: The opening night game was followed by a spectacular opening night fireworks show that was incredibly loud. There are two hotels located just over the left field fence. The game ended about 10:30 p.m. Can you imagine the unexpected jolt that sleeping patrons received? Followed by angry calls to the front desk, I’m sure.
 Huge exhale.
Huge exhale.

